The Indiana Clinical and Translational Institute (CTSI) opened an RFA last year for researchers to make use of the DigiBio data collection platform for their clinical research. Here, we recap the technology and see how the technology has been used to aid research in Indiana.
DigiBio is a HIPAA-compliant application platform which enables secure, remote data collection from research study participants. The platform allows researchers to create surveys, collect data, and utilize features such as audio files and images. DigiBio provides researchers with low-cost personalized research data, and helps keep subjects compliant. If you’re a patient enrolled in a clinical study, using the app, you’ll receive regular notifications to help you keep track of your planned activities, such as surveys, pain-relieving exercises and mindfulness meditations.
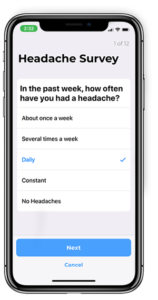 Before DigiBio, researchers had difficulty securely capturing patient-reported outcomes data, and had to use in-person and in-clinic assessments. These assessments were completed by pen and paper, and were inconvenient because they were inconsistent and subjective. DigiBio gives researchers more accurate insights into how a patient is living his or her day-to-day life, and allows researchers to create unlimited personalized app-based studies without spending money on new app development for every study they want to conduct.
Before DigiBio, researchers had difficulty securely capturing patient-reported outcomes data, and had to use in-person and in-clinic assessments. These assessments were completed by pen and paper, and were inconvenient because they were inconsistent and subjective. DigiBio gives researchers more accurate insights into how a patient is living his or her day-to-day life, and allows researchers to create unlimited personalized app-based studies without spending money on new app development for every study they want to conduct.
DigiBio has two different applications: a web app designed for researchers, and a mobile app for study participants. On the web app, researchers are given the opportunity to create studies, enroll participants, and view results from the study that they are currently running. Once the researcher has entered details about his or her participants, those enrolled in the study are given a registration code. Participants only have access to the studies they have been invited to, enabling researchers to run many diverse app-based studies from a single platform.
Indiana CTSI involvement
In 2019, Indiana CTSI recognized that DigiBio’s technology can help researchers capture participant data that was previously too difficult, time consuming, or costly. As a solution, the CTSI issued a new RFA enabling researchers to partner with DigiBio to create their own studies on the platform. Hundreds of Indiana University study subjects have completed studies using the DigiBio app.
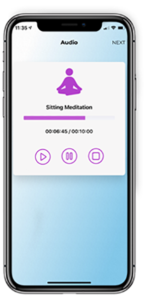
Shelley Johns, PsyD, is an Assistant Professor of Medicine at Indiana University School of Medicine. She wanted to know how much supplementary home meditation practice is needed before a study participant sees a significant improvement in their overall health when facing a medical chronic condition. DigiBio’s flexible application platform allowed her to create and test an app capable of playing guided meditations, tracking participants’ home practice, and collecting immediate effects of each mindfulness practice.
Cian O’Donnell is the Vice President of Business Development at DigiBio. He says Indiana CTSI’s involvement helped to provide a platform for researchers across the country to gather data.
“Indiana CTSI enabled researchers from diverse areas to take advantage of our technology,” said O’Donnell. “Researchers who have used the platform are reaping the rewards, from being able to collect survey data more frequently and easily, to tracking mindfulness practice or capturing pictures from the user’s surroundings.”
Tracking this data provides faster, more accurate results.
DigiBio says it is currently working with researchers from Connecticut to California thanks to the head start provided by the Indiana CTSI.
If you are interested in capturing data in novel ways, email Cian O’Donnell at codonnell@digibiomarkers.com.
Learn additional information or schedule a demonstration here.
